|
Click here to Make your own, heathy version of candies.
Chocolate Peanut Butter Cups from All Day I Dream About Food Mock Payday Bars from Healthy Indulgences Coconut Chocolate Bars from The Nourished Caveman Sugar-Free Twix from All Day I Dream About Food Copycat Ferrero Rocher from Sugar Free Mom Bourbon Chocolate Truffles from I Breathe, I’m Hungry Sugar-Free Gummy Candies from Whole New Mom Better Than Mounds Chocolate Coconut Candies from All Day I Dream About Food Turtles from Maria Mind Body Health Keto Peppermint Patties from Worth Every Chew Sugar-Free Cream Cheese Candies from Low Carb So Simple Dark Chocolate Sea Salt Almond Bark from All Day I Dream About Food Low Carb Chocolate Coconut Fat Bombs from Grassfed Girl Toasted Coconut Butter Cups from Holistically Engineered Low Carb Marshmallows from Keto Diet Blog Whatchamacallit Bars from All Day I Dream About Food White Chocolate Pecan Bark from Carb Wars Pumpkin Fudge from The Coers Family Keto Choconut Almond Butter Cups from The Healthy Foodie Maple Walnut Candy from All Day I Dream About Food Butter Pecan Fat Bombs from Fluffy Chix Cook Strawberry Peeps Fat Bombs from Up Late Anyway Salted Peanut Caramel Clusters from All Day I Dream About Food Snickers Candy Bars from Maria Mind Body Health Lemon Lime Coconut Candies from Delicious Obsessions Salted Caramel Cake Pops from All Day I Dream About Food Reese’s Fudge from Desserts With Benefits Keto Coconut Fat Bombs from Keto Diet Blog Bulletproof Coffee Gummies from Cheerfully Imperfect edit.
0 Comments
In the early 20th century, the German biochemist Otto Warburg believed that tumors could be treated by disrupting their source
of energy. His idea was dismissed for decades — until now. The story of modern cancer research begins, somewhat improbably, with the sea urchin. In the first decade of the 20th century, the German biologist Theodor Boveri discovered that if he fertilized sea-urchin eggs with two sperm rather than one, some of the cells would end up with the wrong number of chromosomes and fail to develop properly. It was the era before modern genetics, but Boveri was aware that cancer cells, like the deformed sea urchin cells, had abnormal chromosomes; whatever caused cancer, he surmised, had something to do with chromosomes. Today Boveri is celebrated for discovering the origins of cancer, but another German scientist, Otto Warburg, was studying sea-urchin eggs around the same time as Boveri. His research, too, was hailed as a major breakthrough in our understanding of cancer. But in the following decades, Warburg’s discovery would largely disappear from the cancer narrative, his contributions considered so negligible that they were left out of textbooks altogether. Unlike Boveri, Warburg wasn’t interested in the chromosomes of sea-urchin eggs. Rather, Warburg was focused on energy, specifically on how the eggs fueled their growth. By the time Warburg turned his attention from sea-urchin cells to the cells of a rat tumor, in 1923, he knew that sea-urchin eggs increased their oxygen consumption significantly as they grew, so he expected to see a similar need for extra oxygen in the rat tumor. Instead, the cancer cells fueled their growth by swallowing up enormous amounts of glucose (blood sugar) and breaking it down without oxygen. The result made no sense. Oxygen-fueled reactions are a much more efficient way of turning food into energy, and there was plenty of oxygen available for the cancer cells to use. But when Warburg tested additional tumors, including ones from humans, he saw the same effect every time. The cancer cells were ravenous for glucose. Warburg’s discovery, later named the Warburg effect, is estimated to occur in up to 80 percent of cancers. It is so fundamental to most cancers that a positron emission tomography (PET) scan, which has emerged as an important tool in the staging and diagnosis of cancer, works simply by revealing the places in the body where cells are consuming extra glucose. In many cases, the more glucose a tumor consumes, the worse a patient’s prognosis. In the years following his breakthrough, Warburg became convinced that the Warburg effect occurs because cells are unable to use oxygen properly and that this damaged respiration is, in effect, the starting point of cancer. Well into the 1950s, this theory — which Warburg believed in until his death in 1970 but never proved — remained an important subject of debate within the field. And then, more quickly than anyone could have anticipated, the debate ended. In 1953, James Watson and Francis Crick pieced together the structure of the DNA molecule and set the stage for the triumph of molecular biology’s gene-centered approach to cancer. In the following decades, scientists came to regard cancer as a disease governed by mutated genes, which drive cells into a state of relentless division and proliferation. The metabolic catalysts that Warburg spent his career analyzing began to be referred to as “housekeeping enzymes” — necessary to keep a cell going but largely irrelevant to the deeper story of cancer. The Health Issue “It was a stampede,” says Thomas Seyfried, a biologist at Boston College, of the move to molecular biology. “Warburg was dropped like a hot potato.” There was every reason to think that Warburg would remain at best a footnote in the history of cancer research. (As Dominic D’Agostino, an associate professor at the University of South Florida Morsani College of Medicine, told me, “The book that my students have to use for their cancer biology course has no mention of cancer metabolism.”) But over the past decade, and the past five years in particular, something unexpected happened: Those housekeeping enzymes have again become one of the most promising areas of cancer research. Scientists now wonder if metabolism could prove to be the long-sought “Achilles’ heel” of cancer, a common weak point in a disease that manifests itself in so many different forms. There are typically many mutations in a single cancer. But there are a limited number of ways that the body can produce energy and support rapid growth. Cancer cells rely on these fuels in a way that healthy cells don’t. The hope of scientists at the forefront of the Warburg revival is that they will be able to slow — or even stop — tumors by disrupting one or more of the many chemical reactions a cell uses to proliferate, and, in the process, starve cancer cells of the nutrients they desperately need to grow. Advertisement Even James Watson, one of the fathers of molecular biology, is convinced that targeting metabolism is a more promising avenue in current cancer research than gene-centered approaches. At his office at the Cold Spring Harbor Laboratory in Long Island, Watson, 88, sat beneath one of the original sketches of the DNA molecule and told me that locating the genes that cause cancer has been “remarkably unhelpful” — the belief that sequencing your DNA is going to extend your life “a cruel illusion.” If he were going into cancer research today, Watson said, he would study biochemistry rather than molecular biology. “I never thought, until about two months ago, I’d ever have to learn the Krebs cycle,” he said, referring to the reactions, familiar to most high-school biology students, by which a cell powers itself. “Now I realize I have to.” Born in 1883 into the illustrious Warburg family, Otto Warburg was raised to be a science prodigy. His father, Emil, was one of Germany’s leading physicists, and many of the world’s greatest physicists and chemists, including Albert Einstein and Max Planck, were friends of the family. (When Warburg enlisted in the military during World War I, Einstein sent him a letter urging him to come home for the sake of science.) Those men had explained the mysteries of the universe with a handful of fundamental laws, and the young Warburg came to believe he could bring that same elegant simplicity and clarity to the workings of life. Long before his death, Warburg was considered perhaps the greatest biochemist of the 20th century, a man whose research was vital to our understanding not only of cancer but also of respiration and photosynthesis. In 1931 he won the Nobel Prize for his work on respiration, and he was considered for the award on two other occasions — each time for a different discovery. Records indicate that he would have won in 1944, had the Nazis not forbidden the acceptance of the Nobel by German citizens. That Warburg was able to live in Germany and continue his research throughout World War II, despite having Jewish ancestry and most likely being gay, speaks to the German obsession with cancer in the first half of the 20th century. At the time, cancer was more prevalent in Germany than in almost any other nation. According to the Stanford historian Robert Proctor, by the 1920s Germany’s escalating cancer rates had become a “major scandal.” A number of top Nazis, including Hitler, are believed to have harbored a particular dread of the disease; Hitler and Joseph Goebbels took the time to discuss new advances in cancer research in the hours leading up to the Nazi invasion of the Soviet Union. Whether Hitler was personally aware of Warburg’s research is unknown, but one of Warburg’s former colleagues wrote that several sources told him that “Hitler’s entourage” became convinced that “Warburg was the only scientist who offered a serious hope of producing a cure for cancer one day.” Although many Jewish scientists fled Germany during the 1930s, Warburg chose to remain. According to his biographer, the Nobel Prize-winning biochemist Hans Krebs, who worked in Warburg’s lab, “science was the dominant emotion” of Warburg’s adult life, “virtually subjugating all other emotions.” In Krebs’s telling, Warburg spent years building a small team of specially trained technicians who knew how to run his experiments, and he feared that his mission to defeat cancer would be set back significantly if he had to start over. But after the war, Warburg fired all the technicians, suspecting that they had reported his criticisms of the Third Reich to the Gestapo. Warburg’s reckless decision to stay in Nazi Germany most likely came down to his astonishing ego. (Upon learning he had won the Nobel Prize, Warburg’s response was, “It’s high time.”) “Modesty was not a virtue of Otto Warburg,” says George Klein, a 90-year-old cancer researcher at the Karolinska Institute in Sweden. As a young man, Klein was asked to send cancer cells to Warburg’s lab. A number of years later, Klein’s boss approached Warburg for a recommendation on Klein’s behalf. “George Klein has made a very important contribution to cancer research,” Warburg wrote. “He has sent me the cells with which I have solved the cancer problem.” Klein also recalls the lecture Warburg gave in Stockholm in 1950 at the 50th anniversary of the Nobel Prize. Warburg drew four diagrams on a blackboard explaining the Warburg effect, and then told the members of the audience that they represented all that they needed to know about the biochemistry of cancer. Source: http://www.nytimes.com/2016/05/15/magazine/warburg-effect-an-old-idea-revived-starve-cancer-to-death.html?_r=0 Low fat diets and exercise are pointless for those wanting to lose weight and obese people should simply eat less, a former shadow health minister told the House of Lords yesterday. Lord McColl, emeritus professor of surgery at Guys Hospital in London, warned that current health advice to avoid fat was ‘false and misleading’ and was fuelling the obesity epidemic.
Speaking at a House of Lords debate, the former surgeon warned that exercising was useless against the huge levels of calories from carbohydrates and sugars that people are now consuming. He warned that the obesity epidemic was as bad for public health as the 1919 flu epidemic. “In the UK the Department of Health and Nice (National Institute for Health and Care Excellence) maintains for many years that the obesity epidemic was due to lack of exercise,” he told peers. “It’s a pity that the 500 people employed by Nice didn’t think to go into the gymnasium get on a machine and exercise to see how few calories you actually burn off. One can pedal away on one of those machines for half an hour and only two or three hundred calories are burned up. One has to run miles to take a pound of fat off. “The whole subject has been bedevilled by all sorts of theories about the course of the obesity; genetics, epigenetic, psychological disturbances. None of them is the cause of the obesity epidemic. One fact remains. It is impossible to be obese unless one is eating too many calories.” In May the National Obesity Forum and the Public Health Collaboration called for a major overhaul of dietary guidelines saying 30 years of urging people to adopt low-fat diets was having ‘disastrous health consequences.’ Their report claimed the low-fat and low-cholesterol message, which has been official policy in the UK since 1983, was based on “flawed science” and had resulted in an increased consumption of junk food and carbohydrates. Lord McColl said eating fat was important because it kept people feeling fuller for longer, and advised overweight people to start adding fat into their diet. “Fat enters the small intestine and greatly delays the emptying of the stomach,” he told peers. “As the stomach emptying is delayed it gives the feeling that one has had enough to eat. Later when the fat has been absorbed the stomach then starts to empty again, It’s a beautifully balance mechanism which tends to prevent us from eating too much and prevents us from getting obese.” Researchers at Imperial College recently found that Britons are on course to be the fattest in Europe within a decade, with almost four in 10 people predicted to be dangerously overweight by 2025. Earlier this week, Sir Simon Stevens the chief executive of the NHS said the obesity crisis was now costing more than the police and fire brigade combined. “Obesity and its related illness is costing the country a fortune and it is not sustainable,” said Baroness Jenkin who called the debate in the Lords. “If we don’t wake up to the extent of this crisis the NHS could end up bankrupt. Already enormous amounts of money are spent on disease which are entirely preventable “The current dietary advice is confusing. The ‘Eat Well guide recommends potatoes, rice, pasta and other starchy carbs. Are we so sure that is good advice? We feed starchy crops to fatten animals so why would they not have the same effect on us?” Health minister Baroness Chisholm said: “There is no point going to an exercise class or a gym then going around the corner for a fizzy drink a donut. It is this sort of culture that needs to change. “Tackling obesity is an important issue. Obesity is a complex issue to which there is no single solution. “I would like to underline that Public Health England bases it dietary guidelines on comprehensive reviews. They consultant with academics, health charities and public health professionals.” Key conclusions | National Obesity Forum report Eating fat does not make you fat Evidence from multiple trials reveals that a higher-fat, lower carbohydrate diet is superior to a low-fat diet for weight loss and cardiovascular risk reduction Stop counting calories Calories from different foods have different metabolic effects on the body, so the cumulative calorie count is meaningless You cannot outrun a bad diet Obesity is a hormonal disorder leading to abnormal energy partitioning, which cannot be solely fixed by increasing exercise Saturated fat does not cause heart disease and full-fat dairy is likely to be protective New meta-analysis of the evidence available forty years ago does not support dietary fat restrictions Avoid at all costs: “processed foods labelled “low fat”, “lite”, “low cholesterol” or “proven to lower cholesterol” No single piece of evidence exists that demonstrates reducing dietary saturated fat reduces cardiovascular events and death Snacking will make you fat The increase in meal frequency plays an equal if not larger role in obesity and has largely been ignored. Source: http://www.telegraph.co.uk/science/2016/06/09/low-fat-diets-and-exercise-are-pointless-for-losing-weight-warns/ Chronic obstructive pulmonary disease or COPD is a lung condition caused by the body’s chronic inflammatory response to inhaled gases and particles. Bacterial infections may also contribute. Accumulated T lymphocytes attract inflammatory white blood cells that release proteases that breakdown of the connective tissue of the lungs. High concentrations of free radicals in tobacco and the chemicals released by inflammatory immune cells damage the cells lining small and large bronchial tubes leading to cell death and scaring or hyper secretion of mucus, hence chronic coughing. Usually, one pathology or the other predominates creating primarily emphysema or chronic bronchitis. Once the process gets started it’s difficult to stop even after quitting smoking.
Cannabidiol from organic hemp could stop this process by several mechanisms. First, CBD suppresses the T cells involved and prevents them from recruiting immune cells that damage the lung. Second, it blocks the inflammatory substances these cells secrete, like Tumor Necrosis Factor and Interleukin-6. Third, it inhibits bronchial tube constriction improving airflow. Fourth, it blocks acute lung injury to prevent the initial inflammatory response using adenosine cellular receptors. And Fifth, CBD increases lung function by decreasing airway resistance so exhaling is much easier. “CBD administered therapeutically, i.e. during an ongoing inflammatory process, has a potent anti-inflammatory effect and also improves the lung function. Case Study: 70 yo retired psychiatrist refinishing his sailboat mixed a stripping and cleaning chemicals that created chlorine gas in the confined space of his galley. He had immediate coughing and shortness of breath, was admitted per ambulance with oxygen support. He immediately started CBD by e-cigarette vaporization. His oxygenation was stable and he was discharged the next day. Cough and chest tightness resolved over the next 3 days with no residual impairment. In this case CBD appears to have halted a potentially serious lung injury and prevented the inflammation that could have lead to extended or chronic disease. Immunopharmacol Immunotoxicol. 2015 Feb;37(1):35-41. Pulm Pharmacol Ther. 2013 Jun;26(3):373-9 Eur J Pharmacol. 2012 Mar 5;678(1-3):78-85. CBD blocks narcotic addiction but now I am talking about alcohol, cocaine, heroin, and meth. CBD inhibits CB1 activation (Thomas, 2007) in the cell, brain and peripheral nerves. The result, from both theory and clinical experience, is the reduction of drug tolerance, addictive behavior and withdrawal. This means less drugs are needed to a...chieve the same benefits, the elimination of the addictive cravings and, very importantly, withdrawal prevention. I also want to add nicotine and benzodiazepines to the list of what I have seen clinically.
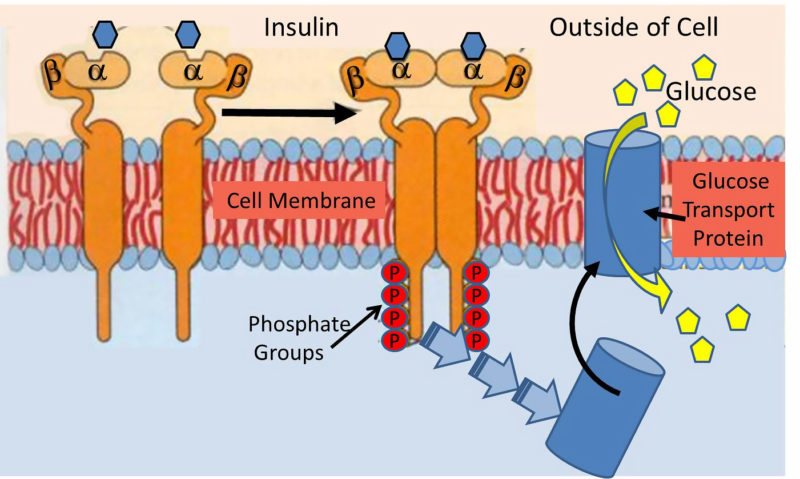
"Studies with drugs of abuse suggested that cannabinoid receptors are involved in the regulation of the central reward system. Accordingly, inactivation of cannabinoid CB1 receptors attenuates the rewarding effects of cannabinoids (Ledent et al, 1999), opiates (Ledent et al, 1999; Martin et al, 2000; Cossu et al, 2001), and cocaine (Chaperon et al, 1998), whereas stimulation of these receptors elicits relapse not only to cannabinoid consumption but also to cocaine, heroin, alcohol, and methamphetamine (Fattore et al, 2007; Higuera-Matas et al, 2008)." (De Chiara, Neuropsychopharmacology, 2010). http://goo.gl/J6T3mk We don't usually think about it but sugar evokes some of the same addictive properties as the worst addictive drugs..."Furthermore, the endocannabinoid system has a key role in the rewarding properties of palatable foods, and cannabinoid CB1 receptor blockade decreases motivation for sweet foods, whereas activation of these receptors increases it." Blair comments: This fact about the attenuation of all addictive compulsions with CB1 inhibition by CBD could lead to the most substantial public health improvement of the century. Study: Voluntary Exercise and Sucrose Consumption Enhance Cannabinoid CB1 Receptor Sensitivity in the Striatum http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3055381/ 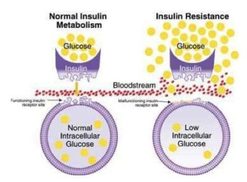 Our current paradigm of insulin resistance is that of a lock and key, and it’s simply wrong. Insulin is a hormone that acts upon a hormonal receptor on a cell surface in order to have an effect. This is often referred to as lock and key model. The lock is the insulin receptor which keeps the gates to the cell closed. When the proper key (insulin) is inserted, then the gate opens to let glucose from the blood inside the cell. This glucose is then able to power the cell machinery. Once you remove the key (insulin) then the gate closes back up and glucose in the blood is no longer able to go inside the cell. Lock and key during insulin resistance What happens during the phenomenon of insulin resistance? Classically, we imagine that the lock and key no longer fit very well. The key (insulin) is able to open the lock (receptor) but only partially and not very well. As a result, the glucose is not able to pass through the gate normally. This results in lower than normal amounts of glucose inside the cell. The glucose, which is now blocked by the closed gate, piles up outside the cell in the blood, which we can detect as elevated blood sugar and make the clinical diagnosis of type 2 diabetes. This has also been described as a state of internal starvation since the cell has little glucose on the inside. The knee-jerk reaction is for the body to increase production of insulin (key). Since each key works less well than previously, the body over-produces the number of keys to make sure that enough glucose goes into the cells. A nice neat theory. The problems The problem, really, is that this paradigm does not really fit reality. First, is the problem the insulin, or the insulin receptor? Well, it’s really quite easy these days to look at the structure of insulin and the structure of the insulin receptor of insulin resistance patients. You simply isolate the insulin or some cells and check their structure with fancy molecular tools. It immediately becomes clear that there is nothing wrong with either the insulin or the receptor. So what’s the deal? The only remaining possibility is that there is something that is gumming up the system. Some kind of blocker that interferes with mechanism of the lock and key. But what? There’s all kinds of theories. Inflammation. Oxidative Stress. Advance glycation End Products. All the usual buzzwords that come out when doctors have really no idea. With this model, we have no real friggin’ idea what caused the insulin resistance. Without understanding what causes IR, we have no chance of treating it. Then there’s the central paradox of hepatic insulin resistance. Let me explain. Insulin has two major actions on the liver. Remember that insulin goes up when you eat. It tells the body to stop producing glucose in the liver (gluconeogenesis) because there is lots of glucose coming in from the stomach (food). This is mediated through the FOX01 pathway. The second major action in the liver is to increase the production of fat (De Novo Lipogenesis (DNL)). This is to deal with the incoming flood of glucose that the body can’t use right way. This is mediated through the SREBP-1c pathway. So, if the liver becomes insulin resistant, then the effect of insulin should drop for both of these actions. That is, the liver should continue to make glucose, and stop making fat. But that’s only the case for gluconeogenesis. That is, during insulin resistance, the liver continues to make new glucose as expected. But DNL (making new fat) continues and actually increases. So insulin’s effect on DNL is not blunted but accelerated! What is going on? How in seven hells can this insulin resistant liver selectively be resistant to one effect of insulin yet accelerate the effect of the other? In the very same cell, in response to the very same levels of insulin, with the very same insulin receptor? That seems crazy. The same cell is insulin resistance and insulin sensitive at the same time! A better explanation: overflow How can we explain this paradox? We need a new paradigm of insulin resistance that better fits the facts. In fact, we can think of insulin resistance as an overflow phenomenon, instead of a lock and key one. All we really know about insulin resistance is that it is much more difficult to move glucose into an ‘insulin resistant’ cell than a normal one. But this does not necessarily mean that the door is jammed. Instead, perhaps the cell is already overflowing with glucose and therefore more glucose cannot go in. Imagine the cell to be a subway car. When the door opens, the passengers on the outside (glucose in the blood) march in a nice orderly manner into the empty subway car (cell). Normally, it doesn’t really require much of a push to get this glucose into the cell (insulin gives the push). But during insulin resistance, the problem is not that the door does not open. The problem, instead is that the subway car (cell) is already overflowing with passengers (glucose). Now the glucose outside the cell simply can’t get in and is left crowded on the platform. Insulin tries to push the glucose into the cell like the Japanese Subway Pushers, but they simply can’t do it because it’s full. So, it looks like the cell is resistant to the effects of the insulin, but really the problem is that the cell is already overflowing. So, the knee jerk reaction is to manufacture more insulin (pushers) to help push glucose into the cell. Which works, but only for a while. So, the cell is not in a state of ‘internal starvation’. Instead, the cell is overflowing with glucose. Glucose starts spilling out into the cell, which looks like gluconeogenesis has not been stopped consistent with insulin resistance. But what happens to fat production? In the classic model of insulin resistance, the paradox was that DNL was enhanced, not decreased which looked a lot like heightened insulin sensitivity instead of resistance. But in the overflow model, the DNL would be enhanced because the cell is trying to rid itself of the excess glucose by producing extra fat. The cell is overflowing and not in an ‘internal starvation’ mode. Why it matters Why is this critically important? Because understanding this new paradigm will lead to the answer of how insulin resistance develops and what we can do about it. The problem does not lie with either insulin nor the insulin receptor. Both are normal. The problem is that the cell is completely stuffed full of glucose. So, what caused it? The answer then seems obvious – it’s a matter of too much glucose and too much insulin. In other words, it was the insulin itself that caused the insulin resistance. We don’t need to chase shadows looking for some mysterious cause of insulin resistance. Once we understand that excessive glucose and excessive insulin is the cause of the insulin resistance, then we can now devise a rational treatment. Reduce insulin and reduce glucose. Once you reverse the insulin resistance, you cure the type 2 diabetes. Source: http://www.dietdoctor.com/a-new-paradigm-of-insulin-resistance |




 RSS Feed
RSS Feed
